INTERNATIONAL CONFERENCE ON HARMONISATION OF TECHNICAL REQUIREMENTS FOR REGISTRATION OF PHARMACEUTICALS FOR HUMAN USE
ICH HARMONISED TRIPARTITE GUIDELINE
BRACKETING AND MATRIXING DESIGNS FOR
STABILITY TESTING OF NEW DRUG SUBSTANCES
AND PRODUCTS
Q1D
Current Step 4 version
dated 7 February 2002
This Guideline has been developed by the appropriate ICH Expert Working Group and has been subject to consultation by the regulatory parties, in accordance with the ICH Process. At Step 4 of the Process the final draft is recommended for adoption to the regulatory bodies of the European Union, Japan and USA.

BRACKETING AND MATRIXING DESIGNS FOR STABILITY TESTING
OF NEW DRUG SUBSTANCES AND PRODUCTS
ICH Harmonised Tripartite Guidelines
Having reached Step 4 of the ICH Steering Committee meeting
on 7 February 2002, this guideline is recommended for
adoption to the three regulatory parties to ICH
TABLE OF CONTENTS
1. INTRODUCTION ................................................................................................ 1
1.1 Objectives of the Guideline............................................................................. 1
1.2 Background ..................................................................................................... 1
1.3 Scope of the Guideline .................................................................................... 1
2. GUIDELINES....................................................................................................... 1
2.1 General............................................................................................................ 1
2.2 Applicability of Reduced Designs ................................................................... 1
2.3 Bracketing....................................................................................................... 2
2.4 Matrixing ........................................................................................................ 3
2.5 Data Evaluation.............................................................................................. 7
BRACKETING AND MATRIXING DESIGNS FOR STABILITY TESTING
OF NEW DRUG SUBSTANCES AND PRODUCTS
1. INTRODUCTION
1.1 Objectives of the Guideline
This guideline is intended to address recommendations on the application of bracketing and matrixing to stability studies conducted in accordance with principles outlined in the ICH Q1A(R) Harmonised Tripartite guideline on Stability Testing of New Drug Substances and Products (hereafter referred to as the parent
guideline).
1.2 Background
The parent guideline notes that the use of matrixing and bracketing can be applied, if justified, to the testing of new drug substances and products, but provides no further guidance on the subject.
1.3 Scope of the Guideline
This document provides guidance on bracketing and matrixing study designs. Specific principles are defined in this guideline for situations in which bracketing or matrixing can be applied. Sample designs are provided for illustrative purposes, and should not be considered the only, or the most appropriate, designs in all cases.
2. GUIDELINES
2.1 General
A full study design is one in which samples for every combination of all design factors are tested at all time points. A reduced design is one in which samples for every factor combination are not all tested at all time points. A reduced design can be a suitable alternative to a full design when multiple design factors are involved. Any reduced design should have the ability to adequately predict the retest period
or shelf life. Before a reduced design is considered, certain assumptions should be assessed and justified. The potential risk should be considered of establishing a shorter retest period or shelf life than could be derived from a full design due to the reduced amount of data collected.
During the course of a reduced design study, a change to full testing or to a less reduced design can be considered if a justification is provided and the principles of full designs and reduced designs are followed. However, proper adjustments should be made to the statistical analysis, where applicable, to account for the increase in sample size as a result of the change. Once the design is changed, full testing or less reduced testing should be carried out through the remaining time points of the stability study.
2.2 Applicability of Reduced Designs
Reduced designs can be applied to the formal stability study of most types of drug products, although additional justification should be provided for certain complex drug delivery systems where there are a large number of potential drug-device interactions. For the study of drug substances, matrixing is of limited utility and bracketing is generally not applicable.
Whether bracketing or matrixing can be applied depends on the circumstances, as discussed in detail below. The use of any reduced design should be justified. In certain cases, the condition described in this guideline is sufficient justification for use, while in other cases, additional justification should be provided. The type and level of justification in each of these cases will depend on the available supporting data. Data variability and product stability, as shown by supporting data, should be considered when a matrixing design is applied.
Bracketing and matrixing are reduced designs based on different principles. Therefore, careful consideration and scientific justification should precede the use of bracketing and matrixing together in one design.
2.3 Bracketing
As defined in the glossary to the parent guideline, bracketing is the design of a stability schedule such that only samples on the extremes of certain design factors (e.g., strength, container size and/or fill) are tested at all time points as in a full design. The design assumes that the stability of any intermediate levels is represented by the stability of the extremes tested. The use of a bracketing design would not be considered appropriate if it cannot be demonstrated that the strengths or container sizes and/or fills selected for testing are indeed the extremes.
2.3.1 Design Factors
Design factors are variables (e.g., strength, container size and/or fill) to be evaluated in a study design for their effect on product stability.
2.3.1.1 Strength Bracketing can be applied to studies with multiple strengths of identical or closely related formulations. Examples include but are not limited to (1) capsules of different strengths made with different fill plug sizes from the same powder blend, (2) tablets of different strengths manufactured by compressing varying amounts of the same granulation, and (3) oral solutions of different strengths with formulations that differ only in minor excipients (e.g., colourants, flavourings).
With justification, bracketing can be applied to studies with multiple strengths where the relative amounts of drug substance and excipients change in a formulation. Such justification can include a demonstration of comparable stability profiles among the different strengths of clinical or development batches.
In cases where different excipients are used among strengths, bracketing generally should not be applied.
2.3.1.2 Container Closure Sizes and/or Fills
Bracketing can be applied to studies of the same container closure system where either container size or fill varies while the other remains constant. However, if a bracketing design is considered where both container size and fill vary, it should not be assumed that the largest and smallest containers represent the extremes of all packaging configurations. Care should be taken to select the extremes by comparing the various characteristics of the container closure system that may affect product stability. These characteristics include container wall thickness, closure geometry, surface area to volume ratio, headspace to volume ratio, water vapour permeation rate or oxygen permeation rate per dosage unit or unit fill volume, as appropriate.
With justification, bracketing can be applied to studies for the same container when the closure varies. Justification could include a discussion of the relative permeation rates of the bracketed container closure systems.
2.3.2 Design Considerations and Potential Risks
If, after starting the studies, one of the extremes is no longer expected to be marketed, the study design can be maintained to support the bracketed intermediates. A commitment should be provided to carry out stability studies on the marketed extremes post-approval. Before a bracketing design is applied, its effect on the retest period or shelf life estimation should be assessed. If the stability of the extremes is shown to be different, the intermediates should be considered no more stable than the least stable extreme (i.e., the shelf life for the intermediates should not exceed that for the least stable extreme).
2.3.3 Design Example
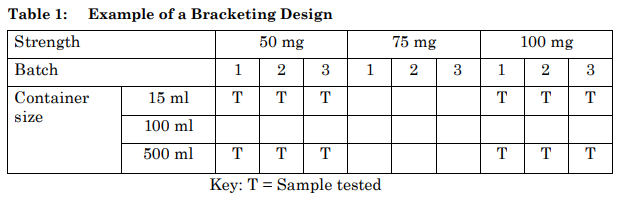
An example of a bracketing design is given in Table 1. This example is based on a product available in three strengths and three container sizes. In this example, it should be demonstrated that the 15 ml and 500 ml high-density polyethylene container sizes truly represent the extremes. The batches for each selected combination should be tested at each time point as in a full design.
2.4 Matrixing
As defined in the glossary of the parent guideline, matrixing is the design of a stability schedule such that a selected subset of the total number of possible samples for all factor combinations would be tested at a specified time point. At a subsequent time point, another subset of samples for all factor combinations would be tested. The design assumes that the stability of each subset of samples tested represents the stability of all samples at a given time point. The differences in the samples for the same drug product should be identified as, for example, covering different batches, different strengths, different sizes of the same container closure system, and possibly, in some cases, different container closure systems.
When a secondary packaging system contributes to the stability of the drug product, matrixing can be performed across the packaging systems.
Each storage condition should be treated separately under its own matrixing design. Matrixing should not be performed across test attributes. However, alternative matrixing designs for different test attributes can be applied if justified.
2.4.1 Design Factors
Matrixing designs can be applied to strengths with identical or closely related formulations. Examples include but are not limited to (1) capsules of different strengths made with different fill plug sizes from the same powder blend, (2) tablets of different strengths manufactured by compressing varying amounts of the same granulation, and (3) oral solutions of different strengths with formulations that differ only in minor excipients (e.g., colourants or flavourings).
Other examples of design factors that can be matrixed include batches made by using the same process and equipment, and container sizes and/or fills in the same container closure system.
With justification, matrixing designs can be applied, for example, to different strengths where the relative amounts of drug substance and excipients change or where different excipients are used or to different container closure systems. Justification should generally be based on supporting data. For example, to matrix across two different closures or container closure systems, supporting data could be supplied showing relative moisture vapour transmission rates or similar protection against light. Alternatively, supporting data could be supplied to show that the drug product is not affected by oxygen, moisture, or light.
2.4.2 Design Considerations
A matrixing design should be balanced as far as possible so that each combination of factors is tested to the same extent over the intended duration of the study and through the last time point prior to submission. However, due to the recommended full testing at certain time points, as discussed below, it may be difficult to achieve a complete balance in a design where time points are matrixed. In a design where time points are matrixed, all selected factor combinations should be tested at the initial and final time points, while only certain fractions of the designated combinations should be tested at each intermediate time point. If full long-term data for the proposed shelf life will not be available for review before approval, all selected combinations of batch, strength, container size, and fill, among other things, should also be tested at 12 months or at the last time point prior to submission. In addition, data from at least three time points, including initial, should be available for each selected combination through the first 12 months of the study. For matrixing at an accelerated or intermediate storage condition, care should be taken to ensure testing occurs at a minimum of three time points, including initial and final, for each selected combination of factors. When a matrix on design factors is applied, if one strength or container size and/or fill is no longer intended for marketing, stability testing of that strength or container size and/or fill can be continued to support the other strengths or container sizes and/or fills in the design.
2.4.3 Design Examples
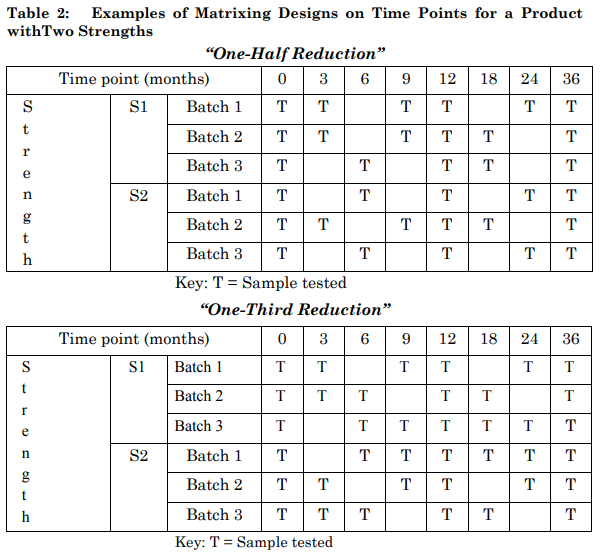
Examples of matrixing designs on time points for a product in two strengths (S1 and S2) are shown in Table 2. The terms “one-half reduction” and “one-third reduction” refer to the reduction strategy initially applied to the full study design. For example, a “one-half reduction” initially eliminates one in every two time points from the full study design and a “one-third reduction” initially removes one in every three. In the examples shown in Table 2, the reductions are less than one-half and one-third due to the inclusion of full testing of all factor combinations at some time points as discussed in section 2.4.2. These examples include full testing at the initial, final, and 12-month time points. The ultimate reduction is therefore less than one-half (24/48) or one-third (16/48), and is actually 15/48 or 10/48, respectively.
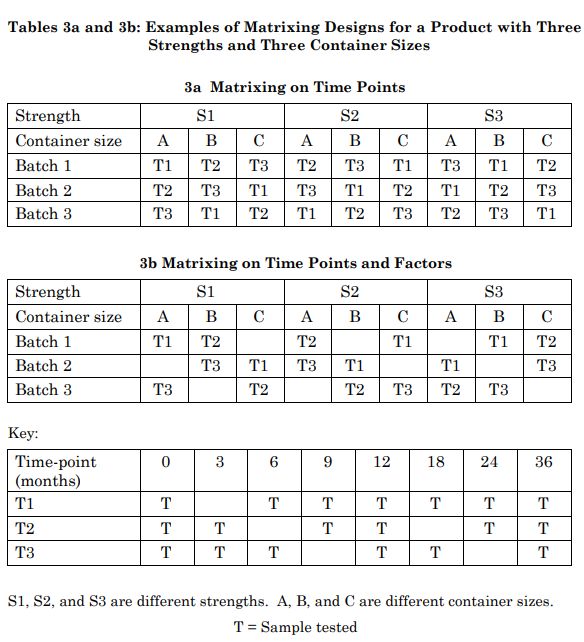
Additional examples of matrixing designs for a product with three strengths and three container sizes are given in Tables 3a and 3b. Table 3a shows a design with matrixing on time points only and Table 3b depicts a design with matrixing on time points and factors. In Table 3a, all combinations of batch, strength, and container size are tested, while in Table 3b, certain combinations of batch, strength and container size are not tested.
2.4.4 Applicability and Degree of Reduction
The following, although not an exhaustive list, should be considered when a matrixing design is contemplated:
knowledge of data variability
expected stability of the product
availability of supporting data
stability differences in the product within a factor or among factors and/or
number of factor combinations in the study
In general, a matrixing design is applicable if the supporting data indicate predictable product stability. Matrixing is appropriate when the supporting data exhibit only small variability. However, where the supporting data exhibit moderate variability, a matrixing design should be statistically justified. If the supportive data show large variability, a matrixing design should not be applied.
A statistical justification could be based on an evaluation of the proposed matrixing design with respect to its power to detect differences among factors in the degradation rates or its precision in shelf life estimation. If a matrixing design is considered applicable, the degree of reduction that can be made from a full design depends on the number of factor combinations being evaluated. The more factors associated with a product and the more levels in each factor, the larger the degree of reduction that can be considered. However, any reduced design should have the ability to adequately predict the product shelf life.
2.4.5 Potential Risk
Due to the reduced amount of data collected, a matrixing design on factors other than time points generally has less precision in shelf life estimation and yields a shorter shelf life than the corresponding full design. In addition, such a matrixing design may have insufficient power to detect certain main or interaction effects, thus leading to incorrect pooling of data from different design factors during shelf life estimation. If there is an excessive reduction in the number of factor combinations tested and data from the tested factor combinations cannot be pooled to establish a single shelf life, it may be impossible to estimate the shelf lives for the missing factor combinations. A study design that matrixes on time points only would often have similar ability to that of a full design to detect differences in rates of change among factors and to establish a reliable shelf life. This feature exists because linearity is assumed and because full testing of all factor combinations would still be performed at both the initial time point and the last time point prior to submission.
2.5 Data Evaluation
Stability data from studies in a reduced design should be treated in the same manner as data from full design studies.
Source from ICH
Copyright © Shijiazhuang Xinfuda Medical Packaging Co., Ltd. All Rights
MAKE AN ENQUIRY